Atomic Makeup
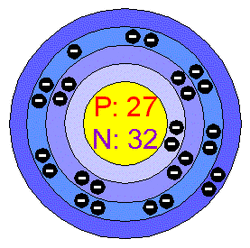
Cobalt's atomic makeup is made up of the atomic number, atomic mass number, the amount of electrons and protons that are in the atom. The atomic number is the number of positive charges or protons in the nucleus of a given atom of an element. The atomic mass number is the number of isotopes in an atom.
Atomic Number: 27Atomic Mass: 58.933195
Electrons: 27
Neutrons: 32
Protons: 27
Atomic Number: 27Atomic Mass: 58.933195
Electrons: 27
Neutrons: 32
Protons: 27
Electron configuration
1s2
2s2 2p6
3s2 3p6 3d7
4s2
2s2 2p6
3s2 3p6 3d7
4s2
Balanced Formula

Cobalt (II) nitrate:
Co(NO3)2 + Na2CO3 --> CoCO3 + 2NaNO3
Co(NO3)2 + Na2CO3 --> CoCO3 + 2NaNO3
Physical Properties

Cobalt is a hard grey metal that resembles iron and nickel. It is only moderately malleable and is fairly ductile. Ductile meaning the capability of being hammered into thin sheets. Cobalt is one of the three naturally occurring magnetic metals along with iron and nickel.
The magnetic properties of Cobalt are extremely obvious in alloys. Alloys are made by melting and mixing two or more metals to create a liquid like substance which then can be formed and shaped into anything that is needed.
The melting point of Cobalt can reach up to at least 1 493 °C. The boiling point of Cobalt is around 3 100 °C. And the density is 8.9 per cubic centimeter.
The magnetic properties of Cobalt are extremely obvious in alloys. Alloys are made by melting and mixing two or more metals to create a liquid like substance which then can be formed and shaped into anything that is needed.
The melting point of Cobalt can reach up to at least 1 493 °C. The boiling point of Cobalt is around 3 100 °C. And the density is 8.9 per cubic centimeter.
Chemical Properties

Cobalt is a moderately active element and combines slowly with oxygen in the air, but it does not catch fire or burn unless it is in a powdered form. Cobalt like most metals reacts to acids to produce hydrogen gas. Cobalt also does not react with water at room temperature.
Phase at room temperature: Solid
Crystal structure: Hexagonal
Period number: 4
Group number: 9
Isotopes: 8
Phase at room temperature: Solid
Crystal structure: Hexagonal
Period number: 4
Group number: 9
Isotopes: 8
